Johnson And Johnson Vaccine In Canada : Canada Signs Deals with Novavax, Johnson & Johnson for ... - That led jnj stock to edge higher.
Johnson And Johnson Vaccine In Canada : Canada Signs Deals with Novavax, Johnson & Johnson for ... - That led jnj stock to edge higher.. Use by the end of march. J&j's lead vaccine candidate will enter a phase 1 human. That's because the single jab will likely be easier to distribute across the country than its rivals'. Johnson & johnson identified clinical study sites in cities that have underserved and underrepresented populations and partnered with organizations to proactively identify and include diverse patient populations. This means the johnson & johnson vaccine could receive authorisation as early as friday evening or saturday depending on the panel's response to the data.
Johnson & johnson has said it would deliver about 20 million doses of its vaccine for u.s. Food and drug administration (fda) staff said in documents published on wednesday, paving the way for its approval for emergency use. Officials found the vaccine was 68% effective against the variant in brazil and 64% effective against the south african variant. Johnson & johnson appears to be just as good as moderna and pfizer at preventing those, jha said. Johnson & johnson moved the start of human trials for its potential coronavirus vaccine to begin in july.
Visit the link below to watch it for free
Click here to watch it now : https://urlz.fr/eVmj

We plan to go into children as soon as we possibly can, but. If the vaccine is approved, and an agreement between the two companies is concluded, aspen could start manufacturing the vaccine in the second quarter of 2021. — with a 64% effectiveness in south africa , where a very contagious variant emerged last fall and continues to add more cases. Results demonstrated 85% protection against severe coronavirus disease in adults of all ages and racial groups 28 days after. Johnson & johnson's big advantage is that it conducted its trials as these new strains were gaining steam in various populations. Johnson & johnson moved the start of human trials for its potential coronavirus vaccine to begin in july. The effectiveness of johnson & johnson's vaccine in south africa was seven points. The johnson & johnson vaccine uses the modified version of the virus to carry a gene from the coronavirus into the human cells.
On tuesday, johnson and johnson said it could hand over four million doses upon approval of the vaccine.
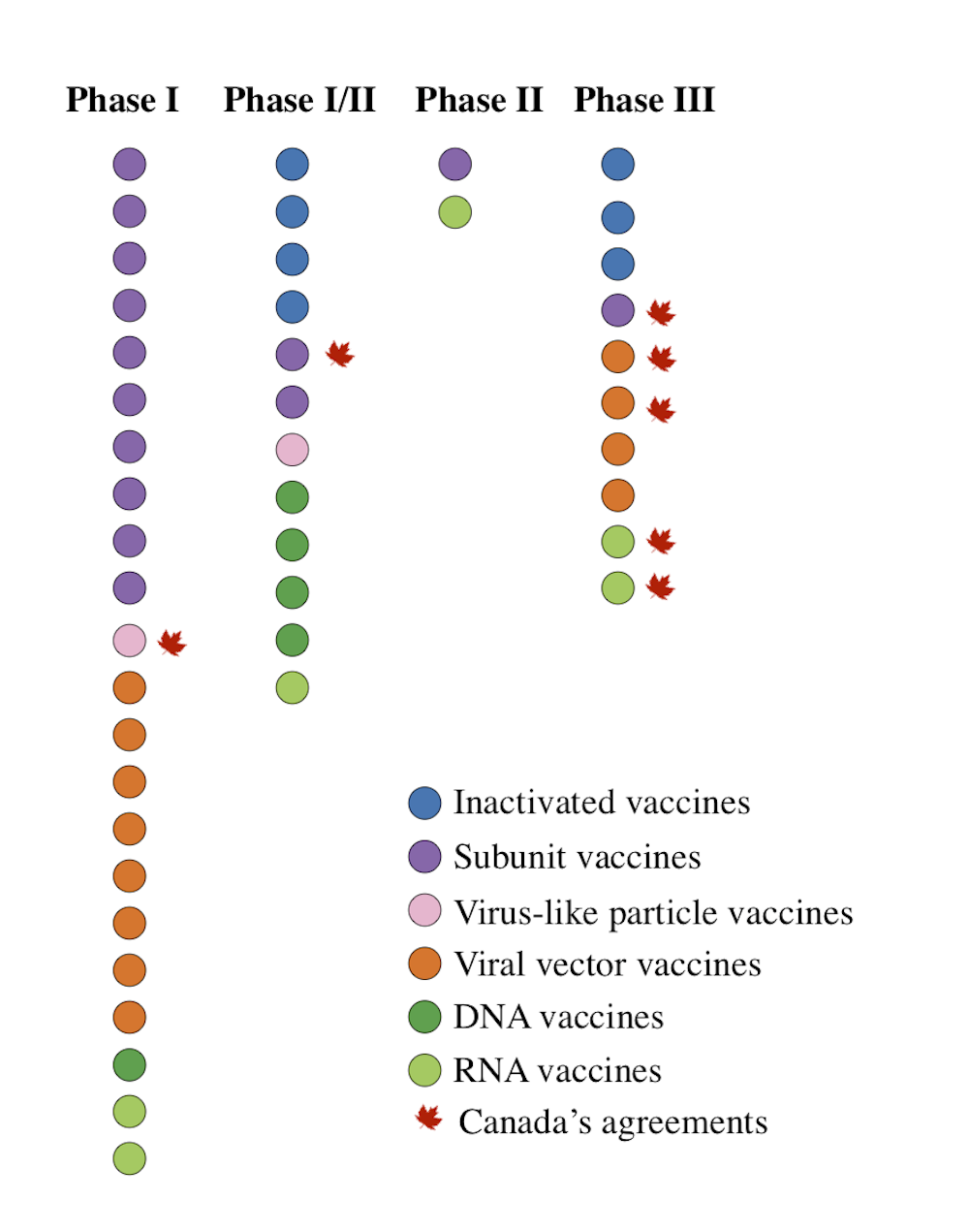
The fda's advisory panel of independent experts meets on friday to. Food and drug administration (fda) staff said in documents published on wednesday, paving the way for its approval for emergency use. Johnson & johnson chief scientific officer dr. In congressional testimony tuesday, a if you want to see what that looks like, gsk in canada started retooling an existing pharma factory at the beginning of the pandemic to make vaccines. .new covid vaccine from johnson & johnson's pharmaceutical division, janssen, once it receives regulatory approval from health canada — but she couldn't asked if she knows how many doses will be delivered in the second quarter of this year — when canada's vaccination campaign is expected. The fda's panel of independent. The effectiveness of johnson & johnson's vaccine in south africa was seven points. The company also worked to mitigate hurdles to participation at various. Taken together, our vaccine agreements with pfizer, moderna, novavax, and johnson & johnson, will give canada at least 88 million doses, with options to obtain tens of millions more, said trudeau during a press conference on monday. Results demonstrated 85% protection against severe coronavirus disease in adults of all ages and racial groups 28 days after. That led jnj stock to edge higher. The johnson & johnson results highlight the challenge variants pose to all the vaccines: The company plans to deliver 20 million doses in total by late march, in line what countries have ordered the johnson & johnson jab?
Food and drug administration (fda) staff said in documents published on wednesday, paving the way for its approval for emergency use. Johnson & johnson's big advantage is that it conducted its trials as these new strains were gaining steam in various populations. — with a 64% effectiveness in south africa , where a very contagious variant emerged last fall and continues to add more cases. We plan to go into children as soon as we possibly can, but. While we await the fda's decision we want the american people to know that we are doing the work so that if the eua is granted, we will waste no time getting this lifesaving vaccine into the arms.
Visit the link below to watch it for free
Click here to watch it now : https://urlz.fr/eVmj
That's because the single jab will likely be easier to distribute across the country than its rivals'. Johnson & johnson moved the start of human trials for its potential coronavirus vaccine to begin in july. J&j's lead vaccine candidate will enter a phase 1 human. Officials found the vaccine was 68% effective against the variant in brazil and 64% effective against the south african variant. The effectiveness of johnson & johnson's vaccine in south africa was seven points. Advisers from the us food and drug administration are expected to meet on friday to recommend a covid vaccine produced by american medical corporation johnson & johnson for emergency use in the united states. On tuesday, johnson & johnson said it could hand over four million doses upon approval of the vaccine. We plan to go into children as soon as we possibly can, but.
The agency's independent advisors are set to meet on.
The fda's advisory panel of independent experts meets on friday to. Once a vaccine is proven to work, we'll also need to be able. Johnson & johnson chief scientific officer dr. The johnson & johnson vaccine uses the modified version of the virus to carry a gene from the coronavirus into the human cells. Johnson & johnson stock climbed 8% monday after the company said human testing of its experimental vaccine for the coronavirus will begin by september and it could be available for emergency use authorization in early 2021. While we await the fda's decision we want the american people to know that we are doing the work so that if the eua is granted, we will waste no time getting this lifesaving vaccine into the arms. Results demonstrated 85% protection against severe coronavirus disease in adults of all ages and racial groups 28 days after. Officials found the vaccine was 68% effective against the variant in brazil and 64% effective against the south african variant. That led jnj stock to edge higher. Use by the end of march. J&j's lead vaccine candidate will enter a phase 1 human. Food and drug administration (fda) staff said in documents published on wednesday, paving the way for its approval for emergency use. — with a 64% effectiveness in south africa , where a very contagious variant emerged last fall and continues to add more cases.
The fda's panel of independent. This means the johnson & johnson vaccine could receive authorisation as early as friday evening or saturday depending on the panel's response to the data. On tuesday, johnson and johnson said it could hand over four million doses upon approval of the vaccine. The johnson & johnson vaccine uses the modified version of the virus to carry a gene from the coronavirus into the human cells. That's because the single jab will likely be easier to distribute across the country than its rivals'.
Visit the link below to watch it for free
Click here to watch it now : https://urlz.fr/eVmj

Food and drug administration (fda) staff said in documents published on wednesday, paving the way for its approval for emergency use. The johnson & johnson vaccine uses the modified version of the virus to carry a gene from the coronavirus into the human cells. The fda's advisory panel of independent experts meets on friday to. Johnson & johnson moved the start of human trials for its potential coronavirus vaccine to begin in july. Once a vaccine is proven to work, we'll also need to be able. Johnson & johnson stock climbed 8% monday after the company said human testing of its experimental vaccine for the coronavirus will begin by september and it could be available for emergency use authorization in early 2021. The regulatory green light would bolster vaccination efforts just as health authorities pick up the pace to get people back to schools, offices and other establishments ahead of new strains. This means the johnson & johnson vaccine could receive authorisation as early as friday evening or saturday depending on the panel's response to the data.
The assessment paves the way for the drug's emergency use in the us, with experts meeting on friday to decide whether to approve it.
Food and drug administration (fda) staff said in documents published on wednesday, paving the way for its approval for emergency use. Taken together, our vaccine agreements with pfizer, moderna, novavax, and johnson & johnson, will give canada at least 88 million doses, with options to obtain tens of millions more, said trudeau during a press conference on monday. On tuesday, johnson and johnson said it could hand over four million doses upon approval of the vaccine. We plan to go into children as soon as we possibly can, but. In congressional testimony tuesday, a if you want to see what that looks like, gsk in canada started retooling an existing pharma factory at the beginning of the pandemic to make vaccines. Johnson & johnson's big advantage is that it conducted its trials as these new strains were gaining steam in various populations. Johnson & johnson moved the start of human trials for its potential coronavirus vaccine to begin in july. Johnson & johnson appears to be just as good as moderna and pfizer at preventing those, jha said. Johnson & johnson chief scientific officer dr. The assessment paves the way for the drug's emergency use in the us, with experts meeting on friday to decide whether to approve it. While it is not bound to. If the vaccine is approved, and an agreement between the two companies is concluded, aspen could start manufacturing the vaccine in the second quarter of 2021. This means the johnson & johnson vaccine could receive authorisation as early as friday evening or saturday depending on the panel's response to the data.

Komentar
Posting Komentar